  
The firm distributed to three medical facilities in Florida, New York and California.ġ A record in this database is created when a firm initiates a correction or removal action. The firm mailed a letter to their customers on Novemstating upgrades will begin November 14, 2003. Support chain attachment on the Bucky carriage may fail and cause the counter weights or X-ray tube to fall. Philips Medical Systems Sales & Service Region No. The firm uses site numbers instead of serial numbers. System, X-Ray, Stationary - Product Code KPR Learn more about medical device recalls.Ģ Per FDA policy, recall cause determinations are subject to modification up to the point of termination of the recall.ģ For details about termination of a recall see Code of Federal Regulations (CFR) Title 21 §7.55.ĥ10(K)s with Product Code = KPR and Original Applicant = PHILIPS MEDICAL SYSTEMS NORTH AMERICA, INC.Class 2 Device Recall Philips Bucky Diagnost FS The record is updated if the FDA identifies a violation and classifies the action as a recall, and it is updated for a final time when the recall is terminated. Worldwide Distribution - US Nationwide and the country of Canada.ġ A record in this database is created when a firm initiates a correction or removal action. In your email, you also clarified that the recalls are to be completed free of charge to any affected customer, whether or not they have a service agreement with Philips Healthcare.įurther questions please call (978) 687-1501. In your March 28, 2013, letter, you proposed a 6 month timeframe for this corrective action, and in your email, you communicated that the field corrections were ongoing and would be completed by July 16, 2013. You have also revised your IATD document to improve instructions to the field engineer for table height measurements. 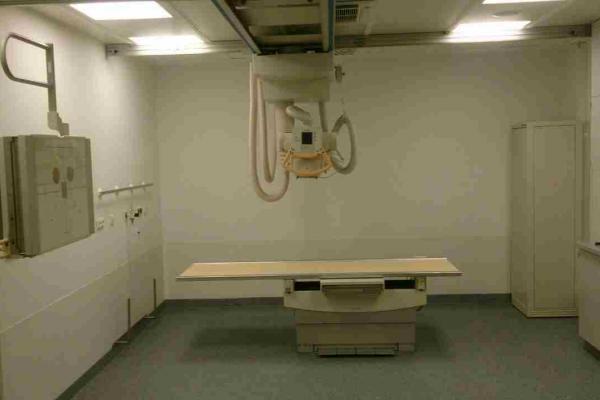
Your proposed corrective action plan (CAP) for the Bucky Diagnost system involves two mandatory field actions including new testing for sites with missing documentation and correcting the table height measurements for systems where the incorrect measurement procedure was used. Field Service Engineer (FSE) will execute corrections and attach record to IATD file. Philips Healthcare issued an Urgent Field Safety Notification on January 11, 2013, to advised users of issued and perform corrective action to the units. Release of Material/Component prior to receiving test results This system is used for making x-ray exposures for diagnostics System, x-ray, stationary - Product Code KPR 
Class 2 Device Recall Philips Bucky Diagnost 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
